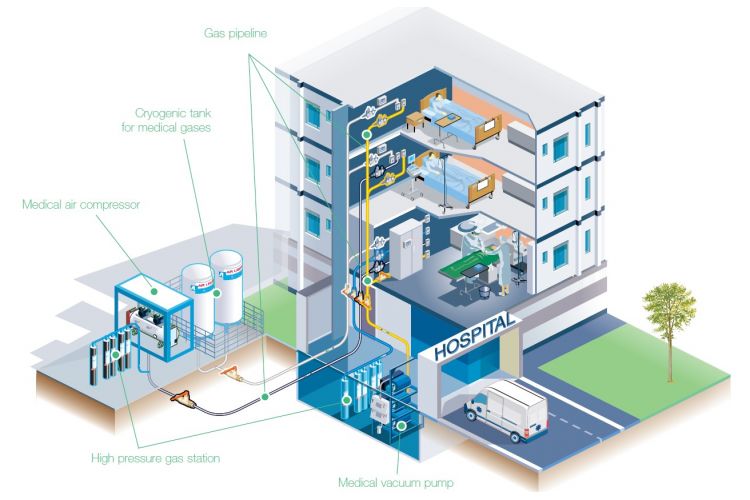
Medical Air is a pharmaceutical-grade, clean, dry, and contaminant-free compressed air used in hospitals and healthcare facilities for respiration and medical procedures.
Medical Air is distributed through the hospital’s medical gas pipeline system (MGPS), similar to oxygen or nitrous oxide.
Production System Components
A typical Medical Air Plant includes:
- Air Compressors
- Filtration Stages (coalescing, activated carbon, and bacterial filters).
- Dryers (desiccant or refrigeration dryers – desiccant is used for medical air).
- Receiver Tanks.
- Monitoring & Control Panel (pressure, dew point, alarms).
- Non-return valves & outlet manifolds connected to the MGPS.
Quality & Purity Specifications:
Medical Air must comply with international standards, primarily:
| Standard | Description |
| ISO 7396-1 | Specifies design, installation, and testing of medical gas pipeline systems. |
| ISO 8573-1:2010 | Defines air purity classes (particles, water, oil). |
| European Pharmacopoeia Monograph 2455 | Defines purity requirements for Medical Air, Compressed. |
Medical Air (Pharmacopoeia / ISO)
| Parameter | Specification / Limit |
| Oxygen content | 20.4 – 21.4 % v/v |
| Carbon monoxide (CO) | ≤ 5 ppm |
| Carbon dioxide (CO₂) | ≤ 500 ppm |
| Nitrogen monoxide (NO) | ≤ 2 ppm |
| Sulphur dioxide (SO₂) | ≤ 1 ppm |
| Water content (dew point) | ≤ –46 °C at 7 bar (or ≤ 67 ppm v/v H₂O) |
| Oil content (total hydrocarbons) | ≤ 0.1 mg/m³ |
| Particulate matter | ≤ 0.1 mg/m³, no particles >40 μm |
| Odor / Taste | None |
| Bacterial contamination | None detectable (sterile after final filtration) |